Create sparkling crystal hearts using pipe cleaners and Borax! Perfect for hands-on, STEM fun in your classroom.
Materials Needed
- Borax powder
- Electric Kettle (or some way to heat water)
- Glass jar or heat-safe container
- Popsicle sticks
- Pipe cleaners
- String
- Tape
- Scissors
- Food coloring (optional)
Instructions:
1. Shape the Hearts
Twist a pipe cleaner into a heart shape. Trim any extra length and double-check that it fits comfortably into your jar. It’s easy to push the pipe cleaner in to start but difficult to pull it out once it’s crystalized.
2. Attach the String
Tie a piece of string to the heart. Tie or tape the other end to a popsicle stick (or pencil) so that the heart hangs freely in the jar without touching the bottom.
3. Prepare the Borax Solution
The ratio of Borax to boiling water is 3:1. Dissolve about 3 tablespoons of Borax per 1 cup of water. Scoop the Borax into the jar, then add the boiled water. You’ll need enough solution to almost fill the jar. Adult supervision is necessary to handle the hot water. Stir until all of the Borax is dissolved.
At this time, you can add optional food coloring and stir until dissolved. Add more than you think! The solution needs to be very dark for the crystals to pick up any color.
4. Submerge the Heart
Lower the heart into the Borax solution so that it hangs without touching the jar’s sides or bottom. Make sure it is fully submerged!
5. Wait for Crystals to Form
Leave the jar in an undisturbed location for 24 hours. No moving the jar around! They need to sit still to work their magic. Crystals will form on the pipe cleaner as the solution cools.
6. Remove and Enjoy
Carefully remove the heart from the solution. Hang it somewhere to dry or place it on a paper towel for a few hours. Your sparkling crystal heart is ready to display!
How It Works
Borax crystals form because the solution becomes supersaturated—meaning it contains more Borax than the water can normally hold at room temperature. As the hot solution cools, the excess Borax begins to come out of solution and attach to the pipe cleaner. Over time, the Borax molecules link together in a repeating pattern, forming the solid crystals you see. This is a great way to explore crystal growth and chemistry in action!



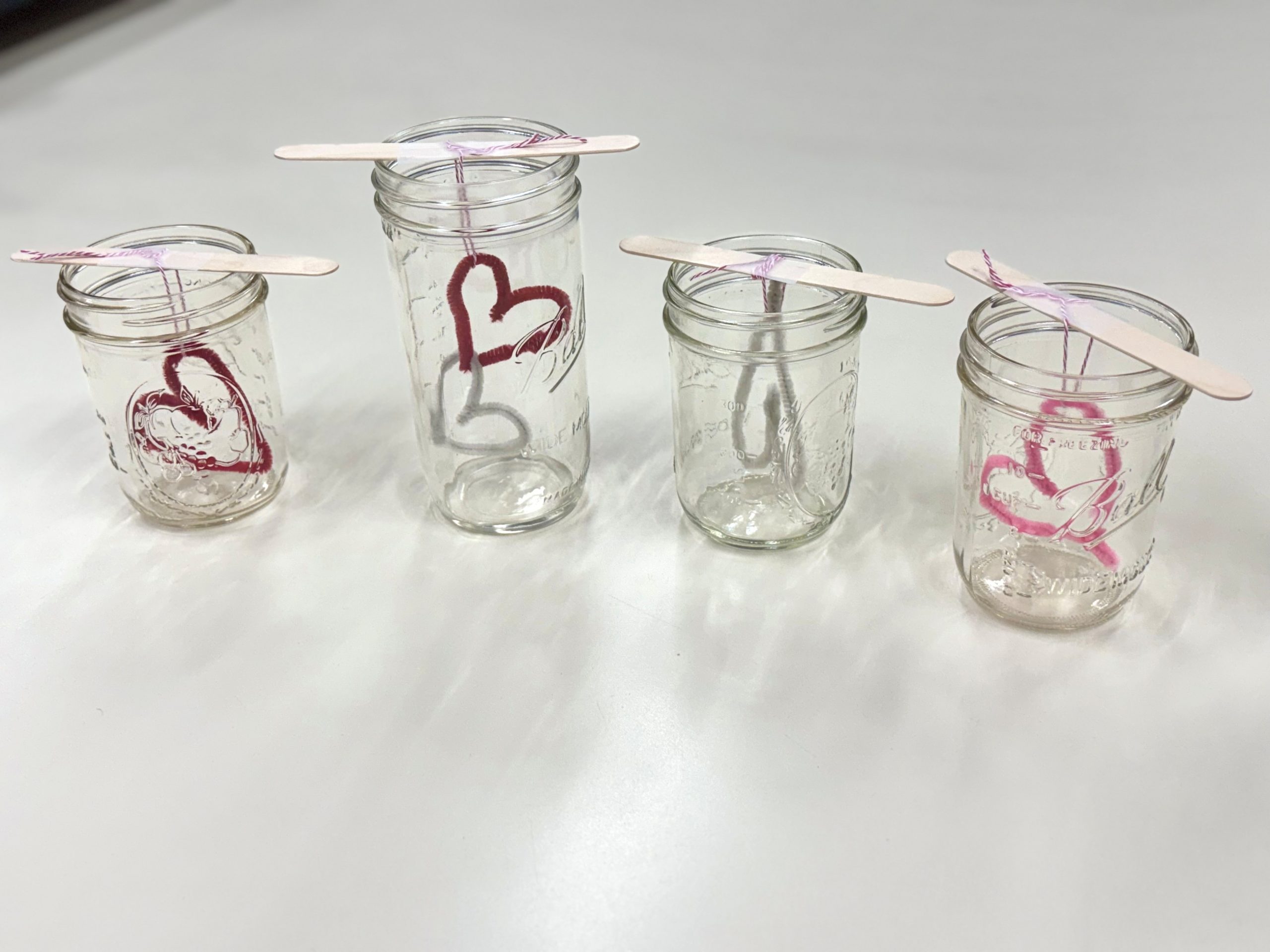











Comments for Borax Crystal Hearts
Add A Comment
Your email address will not be published. Required fields are marked *